Table of Contents
- Introduction: Clarifying a Common Misconception
- Fundamental Fiber Chemistry: The Polyester Structure
- Acid Dyes: Classification and Core Properties
- The Non-Ionic Dyeing Mechanism of Standard Polyester
- Engineering Dyeability: Creating Acid Dye Sites on Polyester
- The Practical “Acid-Dye” Process for Modified Polyester
- Key Influencing Factors and Process Control
- Comparative Analysis: Performance vs. Traditional Dye Systems
- Conclusion and Strategic Outlook
1. Introduction: Clarifying a Common Misconception
For textile professionals in apparel, home furnishings, and industrial manufacturing, dye selection is a cornerstone of product development. The Principle of Dyeing Polyester Filament with Acid Dyes: A Technical and Practical Analysis A frequent point of confusion arises with the statement, “Acid dyes are used for polyester.” Conventionally, this is incorrect. Standard polyethylene terephthalate (PET) polyester is a hydrophobic, non-ionic polymer with no inherent chemical groups (like amino groups in wool or nylon) to form ionic bonds with acid dye anions. Its primary dye class is disperse dyes, which operate through a solid-state diffusion mechanism.
However, the industry landscape is evolving. The core question this article addresses is not about conventional methods, but the scientific and engineered principles that enable polyester filament to be dyed with acid dyes under specific, modified conditions. This knowledge is crucial for product developers seeking unique color effects, enhanced dyeability in blends, or working with next-generation modified polyester fibers. We will deconstruct the chemistry, explore practical applications, and provide a data-driven framework for evaluating this specialized dyeing route.
2. Fundamental Fiber Chemistry: The Polyester Structure
To understand its dyeing behavior, one must start with polyester’s molecular architecture.
- High Crystallinity and Hydrophobicity: PET chains pack tightly, creating a highly ordered, crystalline structure with minimal intermolecular spaces. Combined with its aromatic backbone, this makes polyester inherently water-repellent.
- Lack of Ionic Sites: Unlike protein fibers (wool, silk) or polyamide (nylon), virgin polyester lacks readily ionizable functional groups (e.g., -NH₂) within its polymer chain. Its primary reactive sites are the terminal carboxyl (-COOH) and hydroxyl (-OH) groups, but these are present in very low concentrations and are not cationic under typical dyeing conditions.
- The Dyeing Challenge: This dense, non-polar structure presents a significant barrier to large, water-soluble ionic dyes like acid dyes. Dyeing requires the fiber to “open up,” a process achieved with high temperature (often 120-135°C) for disperse dyes.
3. Acid Dyes: Classification and Core Properties
Acid dyes are water-soluble, anionic colorants primarily designed for fibers with cationic sites. Their classification is based on application pH and bonding mechanism:
Table 1: Classification of Acid Dyes and Their Typical Application
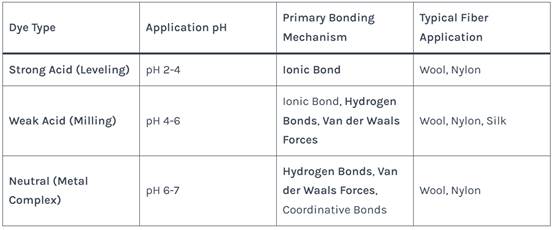
Their molecular size and number of sulfonic acid groups influence solubility and affinity. Weak acid and neutral dyes, with more complex structures and fewer ionic groups, rely more on secondary molecular forces.
4. The Non-Ionic Dyeing Mechanism of Standard Polyester
For standard polyester, the principle of acid dye uptake is not based on ionic attraction. Research indicates it is a minor, secondary process driven by:
- Hydrogen Bonding: Studies, such as on the sorption of Sirius Red 4B, suggest the primary mechanism can be hydrogen bonding between hydroxyl/carbonyl groups on the dye molecule and the carboxyl end groups of the polyester chain.
- Hydrophobic Interactions: The aromatic parts of both the dye and the polyester backbone can interact via hydrophobic forces.
- Limited and Unreliable Uptake: This process is inefficient and produces very pale, non-uniform shades with poor wash fastness, as the dyes are not chemically fixed. Therefore, it is not a commercially viable method for dyeing standard polyester.
5. Engineering Dyeability: Creating Acid Dye Sites on Polyester
To achieve effective acid dyeing, the polyester polymer must be chemically modified to introduce cationic sites. This is the core industrial principle. Two main approaches are:
- Copolymerization: During polymer synthesis, a co-monomer containing a basic amino group (-NH₂) is introduced into the PET chain. This creates a cationic-dyeable polyester (CDP). In an acidic dyebath, these amino groups protonate to form -NH₃⁺, providing sites for ionic bonding with acid dye anions.
- Additive Modification: Masterbatch additives containing basic compounds are blended with standard PET chips before melt spinning. The additive disperses within the fiber, creating localized domains with affinity for acid dyes.
6. The Practical “Acid-Dye” Process for Modified Polyester
When dyeing these modified polyester fibers, the process mirrors that of nylon, leveraging ionic attraction.
- The Ionic Bond Principle: In an acidic bath (typically pH 4-6 for weak acid dyes), the amino groups in the modified fiber acquire a positive charge (Fiber-NH₂ + H⁺ → Fiber-NH₃⁺). The acid dye molecule, as a soluble anion (Dye-SO₃⁻), is electrostatically attracted and forms a strong ionic bond (Fiber-NH₃⁺ ⁻O₃S-Dye).
- The Role of Secondary Forces: Especially with weak acid and neutral dyes, hydrogen bonds and van der Waals forces between the dye and the fiber backbone supplement the ionic bond, improving wash fastness and color yield.
- pH as a Critical Control: pH governs the number of available cationic sites. Lower pH increases protonation and dye uptake speed, but risks unlevel dyeing and fiber damage. Higher pH slows the process, improving levelness. Modern “acid-releasing agents” are used to gradually lower pH during heating, ensuring both levelness and high exhaustion.
- Temperature Profile: The dyebath is heated to 95-100°C to sufficiently swell the polyester matrix, allowing the dye anions to diffuse to the ionic sites.
7. Key Influencing Factors and Process Control
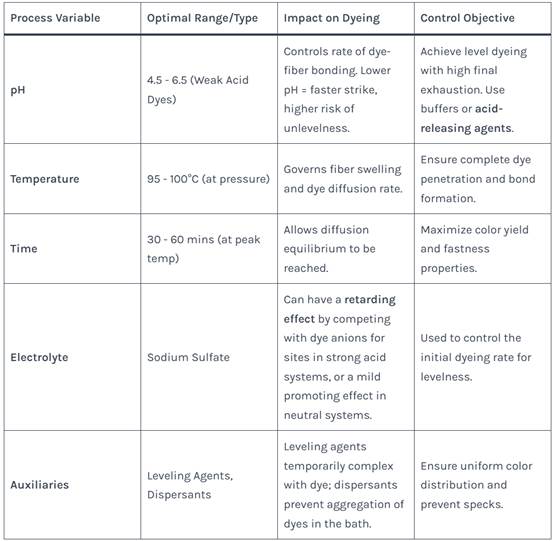
Successful dyeing depends on precise control of several variables.
Table 2: Key Process Variables and Their Impact on Acid Dyeing of Modified Polyester
8. Comparative Analysis: Performance vs. Traditional Dye Systems
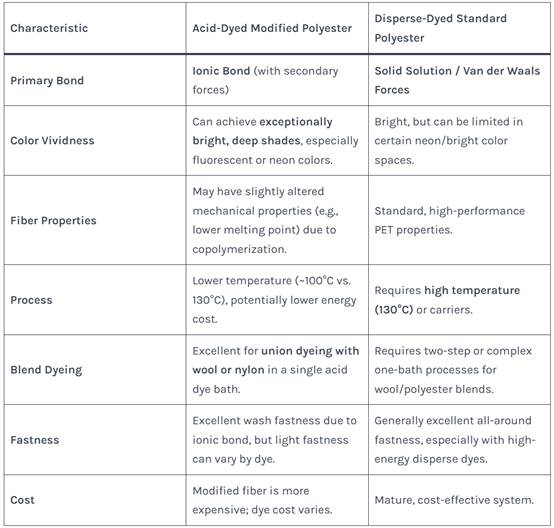
Understanding the trade-offs between acid-dyed modified polyester and standard disperse-dyed polyester is vital for decision-making.
Table 3: Comparison: Acid-Dyed Modified Polyester vs. Disperse-Dyed Standard Polyester
9. Conclusion and Strategic Outlook
The principle of dyeing polyester filament with acid dyes is not a universal method, but a specialized technology enabled by fiber engineering. It shifts the paradigm from physical diffusion to controlled ionic bonding.
For strategic sourcing and product development, this offers distinct advantages:
- Superior Bright Colors: For performance wear, fashion, and plush toys requiring intense hues.
- Simplified Blend Processing: A game-changer for wool/polyester or nylon/polyester blends, simplifying logistics and inventory.
- Sustainability Potential: Lower temperature processing reduces energy consumption.
The future of this technology lies in further refining modified polymers for consistent dye uptake and expanding color palettes. For procurement managers and technicians, mastering this principle opens doors to innovative product differentiation, supply chain simplification, and alignment with evolving market demands for both performance and efficiency. By moving beyond conventional wisdom, it transforms a technical curiosity into a tangible commercial tool.